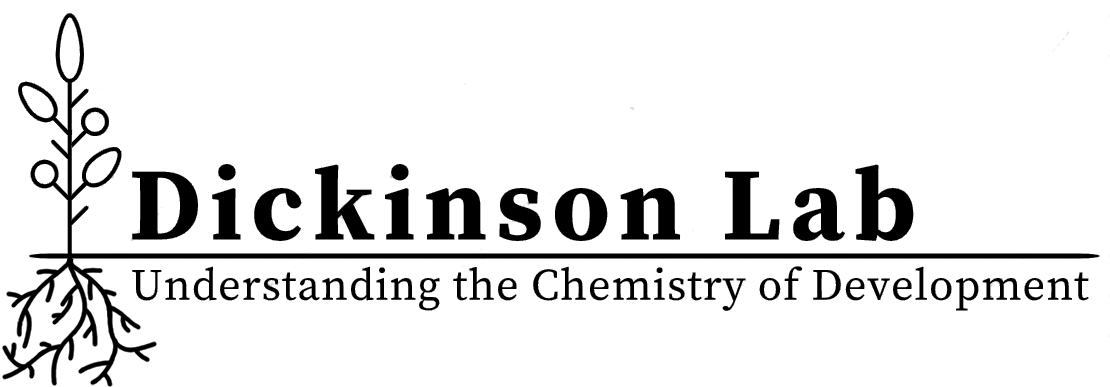Research
Overview
In all multicellular organisms, tissue patterning relies on the ability of stem cells to make the correct decisions—for instance, to divide or differentiate. Small molecules are highly tunable signaling cues, which makes them particularly adept at rapidly transmitting information that guides stem cell decisions. Despite their importance, many small molecule-based mechanisms remain poorly characterized.
We use plant roots to understand how small molecules coordinate stem cell decisions in developing tissue. Roots are an excellent model for finding and monitoring stem cell behavior because root anatomy is arranged along a developmental gradient. Pluripotent stem cells are localized to the meristem, a mitotically active region at the tip of the root. Cell differentiation occurs as cells are displaced towards the shoot. This gradient of differentiation makes it possible to visualize developmental trajectories in a single image.
By integrating this developmental model system with new advances in chemical imaging, our lab explores core principles that govern small-molecule control of stem cell behavior. Widely used methods to visualize gene expression and protein localization have enabled immeasurable progress in understanding cellular mechanisms. Visualization of small molecule activity and localization using approaches such as mass spectrometry imaging could similarly transform our ability to predict and investigate novel biological regulators. Importantly, molecular principles governing small molecule control of stem cell behavior can be conserved between plants and animals, with implications for applications ranging from agriculture to regenerative medicine.
Imaging uncharacterized small molecules along the developmental gradient of the root using mass spectrometry imaging.

Uncovering a new signaling pathway in organogenesis
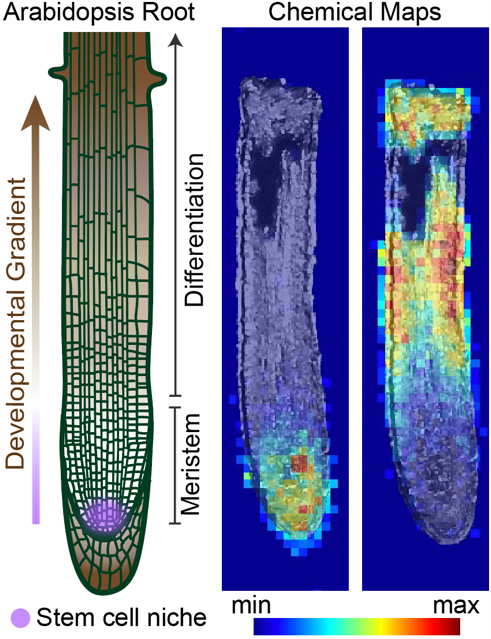
How are organs patterned? The root clock is a dynamic oscillatory process that determines sites of new root organogenesis. By applying chemical genetics, synthetic reporters, and biochemical strategies, we identified retinal and a plant lipocalin as new members of a signaling pathway that regulates the root clock. Dickinson et al, Science, 2021.
A reporter for retinal-binding proteins fluoresces dynamically in the root meristem (the tip of the root that contains actively dividing stem cells) and the region of the root that will form a new site of organogenesis.
Understanding plant development
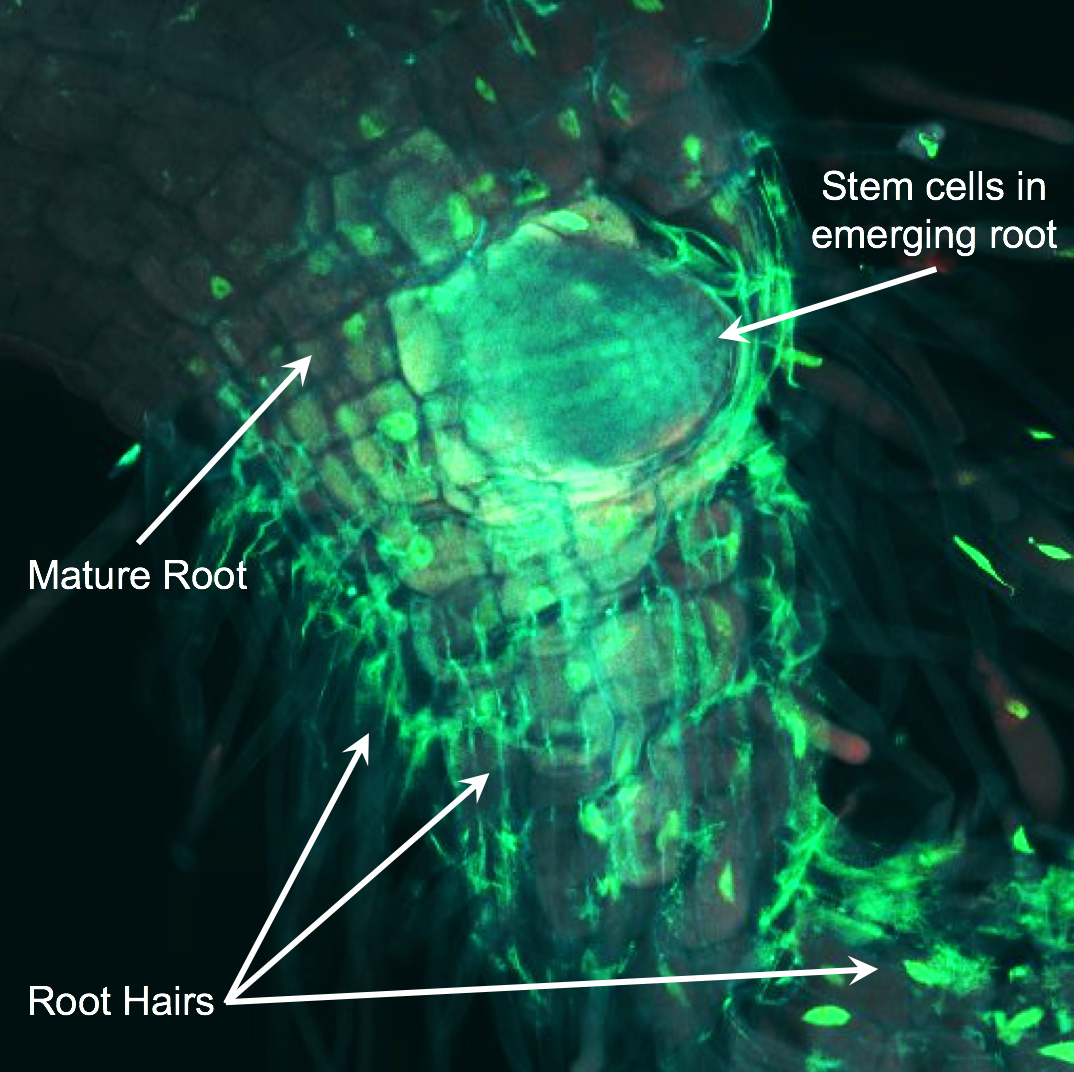
Roots are a powerful model for studying development because there is a developmental gradient along the length of the root. At the tip of the root (the root meristem), stem cells divide to generate progeny that will make up all the cells of the root. Above the meristem, cells elongate and then mature. This highly organized tissue allows us to track stem cell decisions with relative ease.
In the image, a new root is emerging from mature root tissue. GFP is illuminating the nuclei GFP in root cells. Immature stem cells reside at the tip of the root and mature cells (such as root hairs) are closer to the shoot. Jia K.P. et al. Science Advances 2019.
Discovering small molecules that regulate root development
A central question in developmental biology is how the balance between proliferation and differentiation is coordinated during morphogenesis. Due to their mobility, small signaling molecules, such as metabolites, are used by many multicellular organisms to instruct stem cell decisions. Our lab is using a combination of chemical biology, analytical chemistry, and traditional plant biology techniques to discover new small-molecule regulators of development.
These racing roots show the effects of beta-cyclocitrical, a plant hormone that boosts root growth. The rice plants on the left are growing in a gel that contains the hormone, but the ones on the right aren’t getting any help. Dickinson A. J. et al. PNAS 2019